How research in photodynamic therapy may lead to new treatments of deadly diseases like cancer.
Mesothelioma is a form of cancer that is most often associated with asbestos exposure. Despite efforts of national governments such as the United States, Europe, and Japan to ban the use of asbestos due to its toxicity, incidence of mesothelioma or malignant pleural mesothelioma (MPM) remains high at about 3,200 affected individuals per year1. Deaths from the disease has seen a rise within the US, with many of the newly diagnosed cases occurring as a result of non-mining professions, once thought to be the primary culprit for mesothelioma and asbestos exposure.
All said, mesothelioma is a difficult disease to diagnose and treat with a majority of patients unable to survive past a year of diagnosis2. Part of the difficulty in curing the disease lies in the inability to detect it before the late stages, when noticeable symptoms manifest. While much research has been performed to understand the pathways for the cancer to develop, a definitive picture of how asbestos causes the disease on a cellular basis does not yet exist.

SEM Image of Asbestos Fibers. Asbestos remains the lead cause of mesothelioma.
In general, it is understood that asbestos fibers inhaled into the lungs or ingested will penetrate and build up within the mesothelial cells covering the lining of the lungs, heart, or abdomen. From there, they will trigger mutations in the cells and cause inflammation that can lead to cancer. The time between the initial asbestos exposure and diagnosis of the disease may last anywhere from 13 to 70 years. Additionally, many exposed to asbestos may be healthy after a long period of exposure, while others may develop the cancer after only a short period.
Photodynamic Therapy
Cancers diagnosed in their later stages tend to have fewer effective treatment options, contributing to poor survival rates. However, research into new techniques for destroying cancer cells and imaging the disease can shed hope on improving the chances for patients with advanced diseases. One such technique being developed right now is photodynamic therapy, a way to use light to target diseases like cancer.
Photodynamic therapy is a technique that first emerged over a hundred years ago. One of the first demonstrations of using light to treat disease occurred when Niels Finsen discovered that red light exposure could be used to prevent the formation of smallpox, for which he earned the Nobel Prize in 1903. Since then, the concept has evolved over the last 100 years to treat severe acne and reduce tumors, among other applications.
In photodynamic therapy, photosensitizers are introduced to an afflicted region, by injection for example. The photosensitizers traditionally are variants of porphyrins which contain a porphin structure. When the correct wavelength of light is absorbed by the photosensitizer, energy is transferred to oxygen molecules within the photosensitizer resulting in reactive oxygen species (ROS). ROS, sometimes referred to as a kind of “free radical,” can have a number of effects on surrounding biological tissue, including playing a role in cell signaling, gene expression, triggering cell death, and even aging.
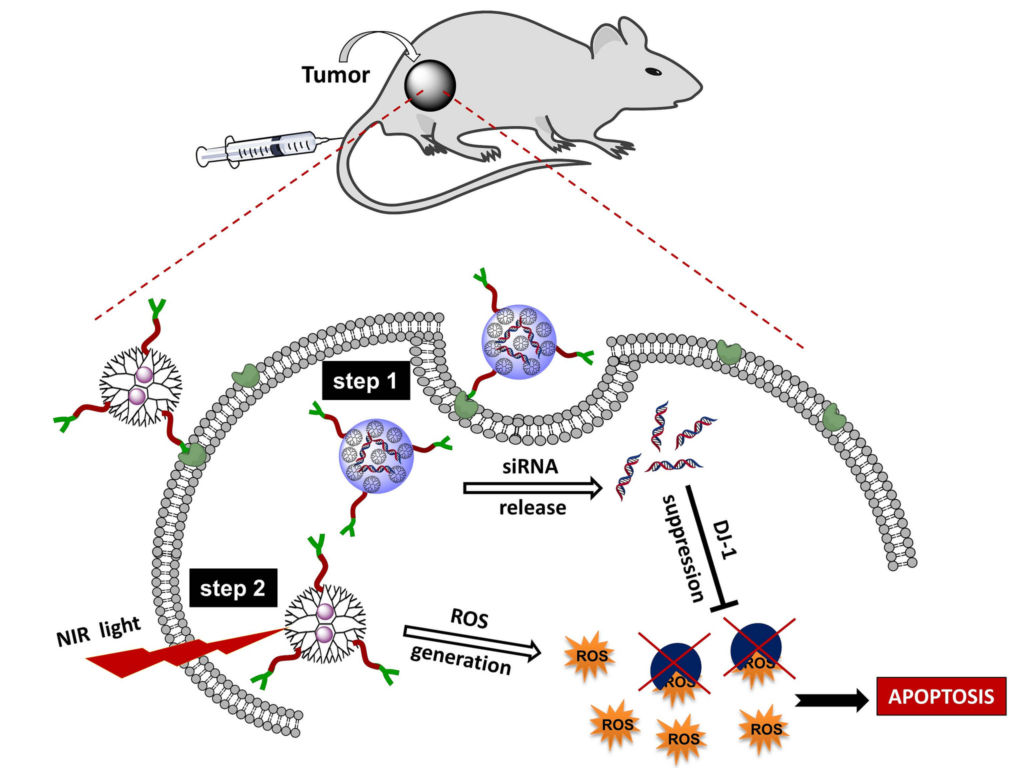
A schematic for one type of photodynamic therapy. Image courtesy of Oregon State University.
The ROS generation of photosensitizers is localized to the area where they reside and is triggered by highly specific wavelengths of light. Therefore, photosensitizers can be placed near cells that are being targeted for treatment, such as malignant tumor cells. This way, the introduction of ROS can be limited to these cells and controlled by the dosage of light input, while avoiding too much harm or unintended side effects to other regions of the body. Since ROS can be harmful to human cells, it is crucial that photodynamic therapy takes advantage of these properties to produce highly specific treatments with minimal side effects.
Photodynamic therapy is a clinically used treatment for destroying tumor cells in patients. Cell destruction can occur by various means. The tumor cell can be directly damaged from light exposure or the presence of the ROS. Similarly, the surrounding tissue that support the tumor cells can be destroyed indirectly leading to the death of the tumor cell. Alternatively, the light generated ROS can trigger an immunoresponse from the body that leads to the destruction of the tumor cell.
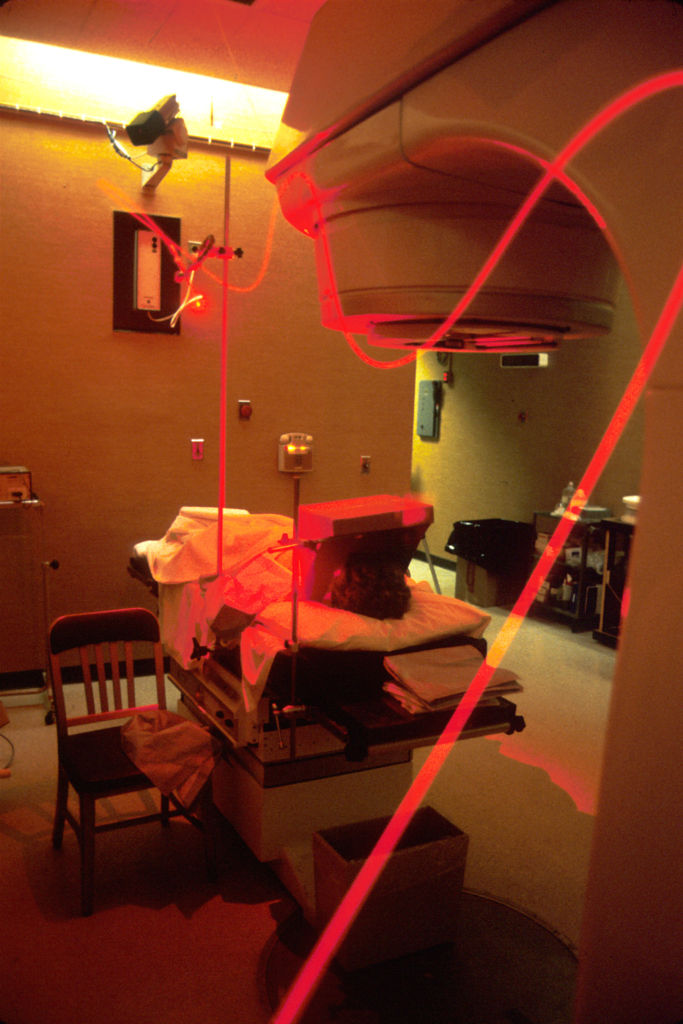
Photograph of a patient receiving photodynamic therapy via fiber optic cable
Nanoparticles as Photosensitizers for Photodynamic Therapy
Advancements in nanotechnology has contributed to new potential photosensitizers capable of advanced photodynamic therapies.
Gold Nanoconjugates
In 2016, a study demonstrated the use of gold conjugate nanoparticles as a photosensitizer to eliminate head and neck cancer cells in mice. Scientists used gold nanoparticles introduced to the tumor cells combined with pulsed infrared laser light to create what they dub a “plasmonic nanobubble.” In this technique, energy from the infrared laser is transferred from the gold nanoparticles into the surrounding tissue or liquid in the form of heat. Adjacent liquids are evaporated resulting in a tiny bubble of vapor that has a lifespan on the order of nanoseconds. The heat exchange from plasmonic nanobubbles are shown to selectively destroy cells in their immediate vicinity.
To selectively attach the nanoparticles to the cancer cells, gold nanoparticles were conjugated to Panitumumab antibodies. The gold-antibody conjugate was injected into mice and subsequently clustered to the cancer cells. In-vivo studies showed the successful elimination of tumor cells while minimizing damage to healthy tissue.
Quantum Dots
Quantum dots, or semiconducting nanocrystals, have been proposed as photosensitizers for their ability to produce highly narrow wavelength light emissions. A recent study has demonstrated the use of graphene based quantum dots as photosensitizers. Two photon excitation of the graphene quantum dots, a nonlinear optical process where a molecule absorbs two photons of the same wavelength simultaneously to reach a higher excitation state, was used to both detect and eliminate multi-drug resistant species of staphylococcus aureus.
Continued development of photodynamic therapy is an exciting field that continues to combine photonics, nanotechnology, chemistry, and biomedical technologies in exciting ways. The advancement of such therapies may lead to new and effective ways to selectively target a number of conventionally hard to treat diseases.
Resources
To read more about how gold nanoparticles can be used in photodynamic therapy, click here.
To read more about how graphene quantum dots are applied to photodynamic therapy, click here.
References
[1] Henley SJ, Larson TC, Wu M, et al. Mesothelioma incidence in 50 states and the District of Columbia, United States, 2003-2008. Int J Occup Environ Health 2013;19:1-10.
[2] Rusch, V.W.; Giroux, D.; Kennedy, C.; Ruffini, E.; Cangir, A.K.; Rice, D.; Pass, H.; Asamura, H.;Waller, D.; Edwards, J.; et al. Initial analysis of the international association for the study of lung cancer mesothelioma database. J. Thorac. Oncol. 2012, 7, 1631–1639.
[3] Lukianova-Hleb, Ekaterina Y., et al. “Intraoperative diagnostics and elimination of residual microtumours with plasmonic nanobubbles.” Nature nanotechnology 11.6 (2016): 525-532.
[4] Kuo, Wen-Shuo, et al. “Antimicrobial Amino-Functionalized Nitrogen-Doped Graphene Quantum Dots for Eliminating Multidrug-Resistant Species in Dual-Modality Photodynamic Therapy and Bioimaging under Two-Photon Excitation.” ACS applied materials & interfaces 10.17 (2018): 14438-14446.
Image Usage
“Cancer Therapy” image courtesy of Oregon State University and used under the Creative commons Attribution-ShareAlike 2.0 Generic (CC BY-SA 2.0) license.
