In ovo spectroscopy is an emerging technology for the welfare of baby chicks. In the egg industry male chicks are considered flaws in production. Egg laying breeds do not grow enough meat for production, and males don’t produce eggs. Traditionally, male chickens were raised for meat and female chickens were kept for laying eggs. However, in the 1920’s-1930’s new breeds emerged that were each efficient in either fattening or egg laying. Thus, making male chick culling a commonplace action.
Today, day old male chicks are often grated, gassed, or even thrown in trash bags and are left to get crushed or suffocate to death. The toll amounts to about 7 billion worldwide per year according to the PoultrySite (about half of new born chicks). Some of the chicks are used in animal feed, fertilizer, or the pharmaceutical industry. However, this accounts for less than 2-3% of the toll. The culling of male chicks, especially on such a large scale raises ethical and animal welfare issues.
Efficiency Concerns
Additionally, the current process of sexing chicks is very labor intensive. Trained workers must sort out subtle differences in wing length or they may have to squeeze out a chick’s feces until its genitalia become visible to determine the chick’s gender. It would be much more convenient for the industry to be able to sort chicks before they hatch. This would save time and money going into incubation and making space for the eggs. Fortunately, new technologies are arising to pre-determine sex of each embryo before hatching. The male eggs can then be used either culled ethically or used in flu vaccines. Flu vaccines require about 100 million fertilized eggs per year.
In today’s article we will explore two different In Ovo Spectroscopy technologies and methods that can be used for chicken egg sex determination.

Day old chicks. Courtesy of KTPRESS.
In Ovo Spectroscopy
Fourier Transform Infrared Spectroscopy
Process
Fourier Transform Infrared (FTIR) Spectroscopy can be used on newly laid chicken eggs. This form of spectroscopy relies on finding the blastoderm and inspecting the chemical makeup of its cells. To start the process of sexing, a device must shine infrared light on the blastoderm. However, the eggshell poses a barrier to optical inspection. Thus, most arising spectroscopic techniques require a small hole inside the eggshell. This can be done very precisely and under one second, with a CO2 laser. If the high-energy laser touches the blastoderm cells, they will be harmed and it could prevent hatching. In order to prevent this occurrence, the laser is directed to only penetrate the calcified shell. This leaves the inner membrane of the shell behind, protecting the future embryo.
Next in the FTIR Spectroscopy process, a detector will collect data on the reflected and transmitted light from the blastoderm cells. This data is a form of “molecular fingerprint” of the cells. Male and female blastoderms contain different chemical molecules, each producing a different light signature. Specifically, male blastoderm cells have more DNA than female cells. This whole sexing process with FTIR Spectroscopy takes only a few seconds.
Effects of In Ovo Spectroscopy- FTIR
In a study done in 2018, the smallest possible hole that would allow optical analysis was 12mm. This study also addressed the affect of FTIR light on hatching rates of inspected eggs. The researchers took a batch of 4,736 eggs and divided them into one control and several experimental group(s). Each of the groups incubation conditions were varied. Shell holes were opened by laser method described above.
The results showed a drastic difference between the hatching rates of non-incubated and incubated eggs post-FTIR Spectroscopy. The longer the eggs were incubated for (up to 72 hrs in this study), the higher the hatching rate. The non-incubated and freshly laid eggs saw a reduction in hatching rate by 6.6% compared to the lowest incubation time (24 hrs). [1] The FTIR process must be done with care as blastoderms are delicate and sensitive to their environment. Infrared light is strong enough to induce molecular changes in the blastoderms.
Lasering of holes for FTIR Spectroscopy. Courtesy of GOTTSCHALK/PHOTOTHEK/BMEL.
Raman Spectroscopy
Raman Spectroscopy is a similar method to FTIR In Ovo Spectroscopy. It is similar in that it utilizes the scattered light pattern of the early embryo cells to determine its biochemical composition. However, Raman Spectroscopy differs from FTIR in the method in which it identifies this composition. One difference is that the light used to illuminate the sample is in the near-infrared (NIR) light range which prevents damaging cells on contact.
Process
To start the Raman scattering process, a diode laser emitting a single wavelength in the NIR range (e.g. 785 nm) should be set up to point at a single blood vessel. It should connect to a microscope using optical fibers in the 100 micrometer range. There are cameras that have been in use that will automatically find and track good blood vessel specimens.
Once the scattering is collected by a detector, the pattern is analyzed through sophisticated methods. For example, chemometric techniques can be used with a superposition of high intensity fluorescent light and the weaker Raman signals. The reason why we cannot just analyze the intensity pattern directly is because there is a lot of scattering variation when Raman spectroscopy is involved. This would lead to inaccurate gender readings.
Analysis
Next, the superposition’s bands can be matched up to the hemoglobin, lipids, and nucleic acids in the blood cells of the embryo. Specifically, the hemoglobin provides the fluorescent light signals. Now we can determine the gender of the chick. As mentioned previously, the sex chromosomes and the amount of DNA in the blood cells of males are 2% larger. Further, after day 13 of incubation male embryos will have a stronger fluorescent signal than females. Additionally, the males have larger signals that correspond to C-C stretching modes and phosphodiester stretching vibrations of the DNA nucleic acids. On the other hand, females will exhibit larger amide III and CHx deformation modes.
Effects of In Ovo Spectroscopy using the Raman method
It should be noted that a 2016 study was done with Raman Spectroscopy where eggs were incubated for 80 to 88 hours. The shell hole width diameter was lasered to as low as 10 mm. Compared to the 12mm study group, the hatching rate was 4% higher. The minimum width of the hole that would allow adequate optical analysis is determined by the highest level of the numerical aperture of the microscope’s objective. However, in this particular study it was found the 12mm holes gave more reliable and consistent results. If the inner membrane covering the hole is punctured, bioadhesive tape may be used to seal it up. [1]
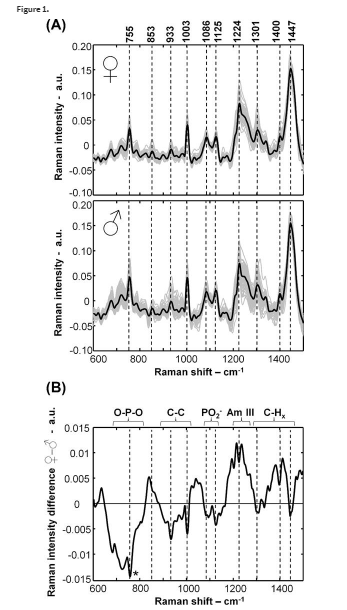
Raman spectra of embryonic chicken blood at d 3.5 of incubation. A: Raman spectra after preprocessing and elimination of the fluorescence signal. B: Difference Raman spectrum obtained by subtracting the mean male spectrum from the mean female one. Courtesy of Galli et al., 2016.
Conclusion
As new technologies emerge utilizing In Ovo Spectroscopy, the mass culling of male baby chicks in cruel ways could soon come to an end. Some concerns about these technologies include the accuracy of sexing, cost-efficiency, and producing fast enough rates for the industry. Further, these techniques should be used before the chick embryo develops its sensory nervous system, which is around day 7, so that culling of the egg does not produce negative reactions. But the future of ethical egg farming is close in sight! The United Egg Producers and Human League are fine tuning these In Ovo Spectroscopic technologies for industrial usage.
Did you know FindLight has a vast collection of FTIR devices and accessories and a series of Raman Spectrometers?

Probably the only way to solve the problem of killing MALE chicks is to INFLUENCE AND CHANGE THE EGG SEX in Embryo.
We are doing it and working hard to arrive at 80-100%.
Do you have reference to that work, might be interesting to add that here…
Hi . I am very concerned about so many male chicks being culled in Australia. Is this technology already being used in Australia? If not when would it be a standard process in Australia. It is very cruel to see them killed this way and have no use for them. Looking forward to your reply.
Michelle Conley
Valid point. Not sure if this is already in practice in Australia. There is some research on that in Germany looks like…
In Germany there are some farmers that are keeping the roosters from being killed and farming them. They can roam outside or in. Roosters have just as much meat as hens and should be farmed the same way as the hens, which would save a lot of money by not employing chicken sexers, making that career obsolete. Hens and Roosters should be processed at least 4 months of age. I find today’s hens don’t have enough meat on them at a measly age of 6 weeks when the poor hens are being slaughtered.
Have commercial Fourier egg desexing machines in other words male and female egg dividers been been developed and marketed yet by any business around the world?