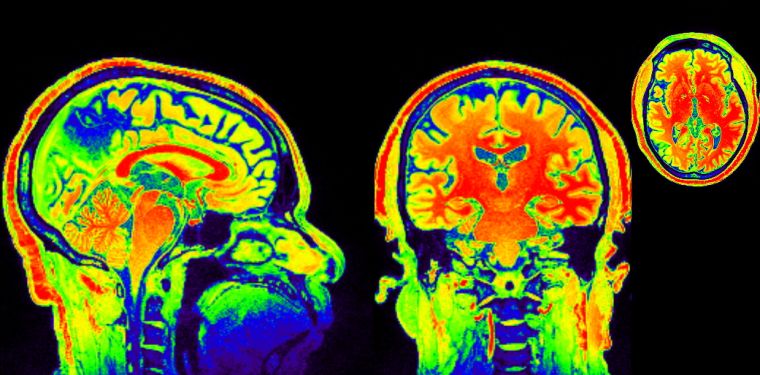
Researchers look to optimize optical coherence tomography by enabling simultaneous bright and dark field (BRAD-OCT) imaging. Courtesy of University of Oxford.
Researchers at the Medical University of Vienna and TU Wien look to optimize optical neuroimaging with their innovative BRAD-OCT technique. Their method, published in the June edition of Biomedical Optics Express, exploits the capabilities of few-mode fibers to provide high-contrast images of abnormal cell growth. Their study could provide great insight for the future of medical research.
The study focuses on two of the most common diseases of the brain: glioblastoma and Alzheimer’s. Alzheimer’s disease often appears as neuritic plaques while glioblastoma takes the form of tumors. According to a report in Neuro-Oncology, glioblastoma is one of the most frequent and the most deadly brain tumors in the United States. 49% of all malignant brain tumors are glioblastoma, and fewer than 6% of patients live five years past their diagnosis date. Researchers hope to learn as much as possible about these diseases by acquiring high-resolution, high-contrast images to characterize the lesions within the brain.
The research team devised a method that combines the proven performance of optical coherence topography (OCT) with the newer field of bright and dark field (BRAD) imaging to produce high-contrast images for in-depth biological research.

An example of bright-field imaging is on the left while dark-field imaging is on the right. Courtesy of Wikipedia.
The Theory Behind BRAD-OCT Imaging
OCT is an imaging technique commonly used in ophthalmology, cardiology, and oncology. It provides cross-sectional 3D images using backscattered signal through tissue. Most OCT setups, however, are limited in scope. Scattering in biological tissue is subject to distinct directionality dependent on the composition of a sample. Standard OCT devices use a single-mode fiber to integrate light directly backscattered by the sample into a small numerical aperture.
This is known as a “bright-field” illumination setup: contrast in the image results from light absorption within the sample. Recent OCT advancements focus on a “dark-field” approach where directly backscattered light is omitted from collection to only collect light scattered by the sample. This provides information about the directionality of the backscatter, therefore adding to overall characterization of the sample. Enabling simultaneous bright and dark field imaging then provides a more complete picture of biological samples.
Introducing the Few-Mode Fiber
The team’s approach is based on OCT interferometers. These setups traditionally use a single-mode fiber to split laser light into a sample arm and reference arm. Both arms travel the same optical distance, but only one will project onto the sample. The beams then meet at a detector to create an interferometric pattern for characterization. This updated system, however, incorporates a few-mode fiber to enable BRAD imaging.
Step-index few-mode fibers allow researchers to account for the directionality of backscattered light from a tissue sample. This is because backscattered light is transmitted along different modes depending on the components’ angular distribution. Different tissues have unique angular backscattering properties, so researchers can use the multiplexed signal to characterize a sample.
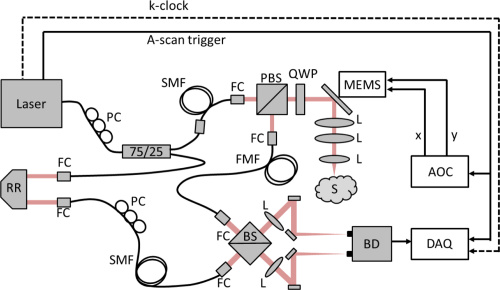
The optical setup for BRAD-OCT is complex, but each device is common in almost any optical lab. Courtesy of OSA Publishing.
The Experimental Setup for BRAD-OCT
The layout of BRAD-OCT imaging is based on that of a Mach-Zender interferometer. A 1310 sweeping laser beam is incident upon series of fibers, lenses, beamsplitters, and waveplates to split, transmit, and create interference with the sample and reference beams. Single-mode fiber optics transfer both arms of the interferometer while the few-mode fiber acts a detection fiber for light backscattered by the sample. The few-mode fiber simultaneously transmits multiple modes at different speeds to guide the light components to the detector unit. Backscattered components, each with distinct directions and modes, interfere with the reference beam to reconstruct modal images at different depth positions. The result is a cross-sectional OCT image of the brain tissue.
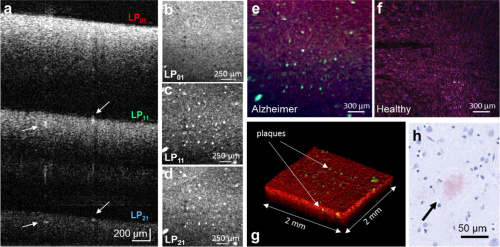
Above images a-f of neuritic plaques portray the vast amount of information acquired through BRAD-OCT: (a) depth-multiplexed images corresponding to higher modes (b) bright-field scattering (c-d) dark-field contrast (e-f) images of Alzheimer’s-afflicted versus healthy brain, respectively
The Results obtained with BRAD-OCT
Angular scattering is directly correlated to particle size. Using the few-mode fiber, the researchers were able to establish a ratio of the directly backscattered light collected by the fundamental mode (LP01) to the light scattered at greater angles collected by higher modes (LP11, for example). LP01 and LP11 (etc.) represent bright field and dark field OCT detection, so the team deemed this angular scattering parameter the “BRAD ratio.”
The research team used their BRAD-OCT setup to image brain tissue samples from glioblastoma and Alzheimer’s patients. These trials were executed to determine the effectiveness of BRAD-OCT in detecting scattering from tumors and neuritic plaques respectively. The team also imaged tissue from a healthy human brain for comparison.
The paper first discusses the 3D image and tissue scattering information acquired from the sample afflicted with glioblastoma. The BRAD-OCT images exhibit a difference in the “scattering signature” when scanning over a tumor, and healthy tissue areas yield a low BRAD ratio. The system also showed the average nuclear size was significantly larger in tumor areas, as to be expected. Next the researchers examined BRAD contrasts in images of neuritic plaques. The plaque structure produces an angular scattering pattern different from the surrounding gray matter. Using the angular detection differences in the few-mode fiber, the team accurately detected and localized these structures. BRAD-OCT is a success.
Utilizing few-mode fibers to create additional channels for light transmission proves to greatly improve sensitivity of the traditional OCT system. BRAD-OCT incorporates the dimension of directionality to provide a high-contrast, high-resolution 3D image. The team believes their method of optical characterization could improve OCT applications beyond neuroimaging. Several specialties in medicine could soon benefit from the enhanced abilities of BRAD-OCT.
